Author associations: Medical Research Council—University of Glasgow Centre for Virus Research, Glasgow, Scotland, UK (G.B. Tyson, S. Jones, N. Logan, P.R. Murcia, B.J. Willett, M.J. Hosie); University of Glasgow, Glasgow (G.B. Tyson, S. Jones, M. McDonald, L. Marshall, W. Weir)
The World Organisation for Animal Health reported that 26 various animal types had actually been contaminated with SARS-CoV-2 by December 31, 2022; ≈30% (8/26) of the vulnerable types are felids (1). Animal SARS-CoV-2 infections stemming from anthropogenic transmission can cause onward animal-to-animal transmission, as explained formerly in mink (2), hamsters (3), and white-tailed deer (4). There have actually likewise been reports of animal-to-human transmission of SARS-CoV-2 from farmed mink (2), family pet hamsters (5), free-ranging white-tailed deer (6), and an animal cat (7).
It is unidentified whether private SARS-CoV-2 variations are basically most likely to be transferred from people to cats or whether contaminated cats are basically most likely to establish medical indications. The goal of this research study was to evaluate the seroprevalence of SARS-CoV-2 infection in cats throughout April 2020–February 2022 in the United Kingdom. We utilized a pseudotype-based neutralization assay (PVNA) to determine infection reducing the effects of antibody titers and a confirmatory ELISA that determined antibodies acknowledging the receptor binding domain of the SARS-CoV-2 spike (S) protein. We determined reducing the effects of titers versus a panel of viral pseudotypes based upon a lentiviral (HIV) foundation and bearing the S proteins of the primary distributing variations in the United Kingdom to examine the uniqueness of the reducing the effects of action. The University of Glasgow Veterinary Ethics Committee given approval for the research study (EA27/20).
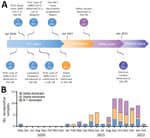
We evaluated recurring blood samples from 2,309 cats by utilizing PVNA at a last dilution of 1:100; the samples were sent to the University of Glasgow Veterinary Diagnostic Services lab (VDS) throughout April 2020–February 2022 (Figure 1, panel A). The samples represented a mate that was broadly representative of the domestic cat population in the United Kingdom, consisting of samples from 112 of the 126 UK postal code locations (Appendix 1 Figure 1), although the samples had an unequal circulation unassociated to the regional human population density. Overrepresented locations consisted of Blackpool, Glasgow, Edinburgh, and Cambridge. The PVNA utilized HIV (SARS-CoV-2) pseudotypes bearing S proteins of SARS-CoV-2 ancestral D614G (B.1), Alpha (B.1.1.7), Delta (B.1.617.2) or Omicron (BACHELOR’S DEGREE.1). Samples sent early in the pandemic were checked versus ancestral D614G (B.1) just, whereas brand-new variations were consisted of as they emerged (Appendix 2). We approximated neutralization titers for positive samples by carrying out the PVNA with serially watered down samples.
Our results revealed that SARS-CoV-2 seroprevalence in UK cats increased with time (Figure 1, panel B). Overall, the seroprevalence throughout the research study duration was 3.2% (95% CI 2.56%–4.05%; 75/2,309). Seroprevalence was greatest throughout September–November 2021 (5.3%, 95% CI 3.69%–7.23%; 35/666) and throughout December 2021–February 2022 (5.2%, 95% CI 3.09%–8.05%; 18/348).
When we examined private samples, we observed distinctions in variant-specific strengths amongst titers versus the various SARS-CoV-2 variations: 17/75 (22.7%) samples were B.1 dominant (i.e., they had greater titers versus B.1 than versus other variations); 31/75 (41.3%) were Alpha dominant, and 27/75 (36%) were Delta dominant. On average, Delta-dominant samples showed greater neutralization titers (suggest 760) versus their dominant pseudotype compared to Alpha-dominant (488; p = 0.06) or B.1-dominant (329; p = 0.02) samples (Appendix 1 Figure 2). Throughout the research study duration (April 2020–February 2022), no Omicron-dominant seropositive samples were recognized; we expected this finding due to the fact that just a little percentage of samples were gathered after the Omicron alternative emerged.
We observed an association in between the dominant version in cats and the timeline of alternative introduction in the human population. Detection of brand-new dominant variations in cats routed detection of the version in the people; nevertheless, we spotted dominant titers versus extinct variations even after human cases had actually decreased, potentially suggesting lasting humoral resistance (Figure 2). We observed 3 unique patterns of neutralization. B.1-dominant samples typically had a little lower titers versus the Alpha pseudotype than versus B.1. Those samples likewise had considerably lower titers versus both the Delta (p<0.0001) and Omicron (p<0.001) pseudotypes. Alpha-dominant samples revealed a little lower B.1 titers and considerably lower Delta and Omicron titers. Delta-dominant samples revealed comparable titers versus the B.1, Alpha, and Omicron pseudotypes, all of which were considerably lower than their Delta titers (p<0.0001) (Appendix 1 Figure 3).
The patterns we observed for cats believed to have actually been contaminated with the B.1 version resemble the patterns of neutralization in people reported formerly (8); Wilhelm et al. revealed that people immunized with an ancestral pressure–based vaccine establish lower neutralization titers versus the Delta and Omicron variations than versus B.1 or Alpha. Another research study revealed that cats experimentally inoculated with either the ancestral or the Delta alternative ended up being sluggish and pyrexic, whereas Omicron-inoculated cats did not establish any medical indications and showed lower levels of infection shedding, recommending that the Omicron alternative may be less pathogenic in cats in addition to in people (9).
Despite those unique patterns of neutralization, the alternative to which the animal was exposed can just be speculatively presumed through serologic screening in the lack of viral series information, even in cases in which the titer versus the dominant version is lot of times higher than the next greatest titer. The 3 particular patterns of resistance we observed resembled previous findings in people (10). It is most likely that both the antigenicity of the various variations’ S proteins and the viral load throughout the infection duration affect the breadth and effectiveness of variant-specific neutralization.
A higher percentage of pure-blooded cats (31/720 [4.3%, 95% CI 2.94%–6.06%]) than nonpedigree cats (39/1,300 [3%, 95% CI = 2.14%–4.08%]) were seropositive; nevertheless, this finding was not considerable (p = 0.1). Purebred cats are most likely to be kept inside just and might for that reason experience more close contact with their owners, indicating they are more susceptible to direct exposure to SARS-CoV-2 if their owners end up being contaminated.
Although a conclusive protective limit antibody level for SARS-CoV-2 has actually not yet been developed, subsiding reducing the effects of antibody levels in people after vaccination have actually been connected with reinfection and minimized security versus unique variations (11). Sequential samples >12 days apart were gathered from 5 seropositive cats. In all 5 cases, the reducing the effects of titers versus SARS-CoV-2 subsided with time. The typical portion reduction in titer each day was extremely variable throughout samples, although for 3 of 5 cats it corresponded throughout all variations (Table).
This research study showed increasing seroprevalence of SARS-CoV-2 antibodies in the UK domestic cat population, constant with outcomes reported in a study of cats and dogs just recently performed in Canada (12) and the low seroprevalence observed throughout the very first and 2nd waves of the pandemic (13,14). This boost might be explained by the determination of the humoral action with time, with a following build-up in the variety of seropositive lead to the population. In addition, increased seroprevalence throughout the later months of the pandemic might suggest the probability of human-to-cat transmission is higher for more recent variations that have actually formerly been revealed to be quicker transferred in between people (15), although this hypothesis has actually not been validated experimentally.
This research study shows the value of embracing a One Health technique to keep track of SARS-CoV-2 infections in family pet cats that remain in close contact with their SARS-CoV-2–positive owners. Changes in transmissibility of emerging variations must be kept an eye on in cats in addition to people.
This post was preprinted at https://www.biorxiv.org/content/10.1101/2022.11.18.517046v1.
Miss Tyson is a PhD prospect at the MRC-University of Glasgow Centre for Virus Research, Glasgow, Scotland. Her main research study interests consist of viral immunology, humoral resistance, and infections at the human–animal user interface.
Top