
Figure 1
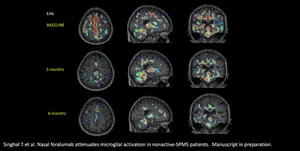
-
Five out of six sufferers in FDA licensed Expanded Access Program are displaying a qualitative discount in microglia activation (a key biomarker being noticed)
-
Foralumab to advance into Phase 2 human medical trials utilizing the world’s solely totally human intranasal anti-CD3 monoclonal antibody
-
Phase 2 trial screening for na-SPMS to begin in November 2023
NEW YORK, Oct. 13, 2023 (GLOBE NEWSWIRE) — Tiziana Life Sciences Ltd. (Nasdaq: TLSA) (“Tiziana” or the “Company”), a biotechnology firm growing breakthrough immunomodulation therapies through novel routes of drug supply, immediately introduced {that a} discount in activated microglia, as seen in six-month Positron Emission Tomography (PET) scans, has now been noticed in a complete of 5 of the six sufferers with non-active secondary-progressive a number of sclerosis (na-SPMS) handled with intranasal foralumab in its Expanded Access Program (EAP). Activated microglia are believed to play a outstanding position within the pathogenesis of neuroinflammatory and neurodegenerative illnesses together with a number of sclerosis, Alzheimer’s illness, and amyotrophic lateral sclerosis, or ALS.
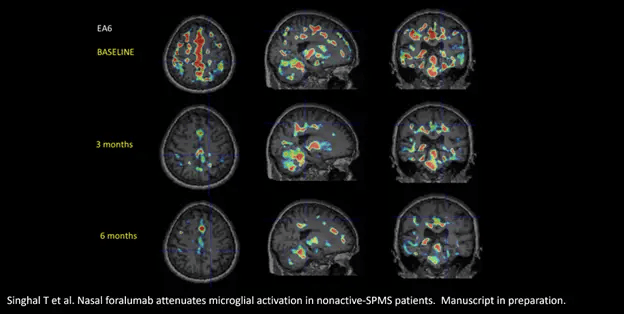
Tarun Singhal, M.B.B.S., M.D., Director of the PET Imaging Program in Neurologic Diseases, affiliate neurologist and nuclear drugs doctor at Brigham and Women’s Hospital, a founding member of Mass General Brigham Healthcare System, and Assistant Professor of Neurology at Harvard Medical School, commented, “Upon review of the baseline and six-month PET scans of the latest cohort of four Expanded Access patients, three out of the four scans suggested a qualitative reduction in the microglial PET signal. When combined with my assessment of the first two Expanded Access patients at six-months, five of the six suggested a reduction in qualitative microglial PET signal. An example of this can be seen in the graphic below, titled, “Figure 1”, displaying the deactivation of this sign in affected person EA6. This is promising from an imaging standpoint, and additional research are wanted to substantiate these findings utilizing further quantitative approaches.”
Howard L. Weiner, M.D., Chairman of Tiziana’s Scientific Advisory Board and Co-Director of the Ann Romney Center for Neurologic Diseases at Brigham and Women’s Hospital added, “With six patients now dosed in our na-SPMS EA program, I feel that Dr. Singhal’s readout of the six-month PET scans strongly supports our previously announced 3-month clinical findings.”
Gabriele Cerrone, Chairman, performing CEO and founding father of Tiziana Life Sciences commented, “I believe that the six-month qualitative na-SPMS PET readout by Dr. Singhal is very encouraging and will enable us to rapidly advance foralumab in Phase 2a testing to address patients afflicted with this devasting disease who currently have no FDA-approved treatments available.”
Figure 1.
About Foralumab
Activated T cells play an essential position within the inflammatory course of. Foralumab, the one totally human anti-CD3 monoclonal antibody (mAb), binds to the T cell receptor and dampens irritation by modulating T cell perform, thereby suppressing effector options in a number of immune cell subsets. This impact has been demonstrated in sufferers with COVID and with a number of sclerosis, in addition to in wholesome regular topics. The non-active SPMS intranasal foralumab Phase 2 trial is predicted to start out screening in November of 2023. Immunomodulation by nasal anti-CD3 mAb represents a novel avenue for therapy of neuroinflammatory and neurodegenerative human illnesses.1,2
About Tiziana Life Sciences
Tiziana Life Sciences is a clinical-stage biopharmaceutical firm growing breakthrough therapies utilizing transformational drug supply applied sciences to allow various routes of immunotherapy. Tiziana’s progressive nasal strategy has the potential to offer an enchancment in efficacy in addition to security and tolerability in comparison with intravenous (IV) supply. Tiziana’s lead candidate, intranasal foralumab, which is the one totally human anti-CD3 mAb, has demonstrated a positive security profile and medical response in sufferers in research to this point. Tiziana’s know-how for various routes of immunotherapy has been patented with a number of functions pending and is predicted to permit for broad pipeline functions.
For additional inquiries:
Tiziana Life Sciences Ltd
Paul Spencer, Business Development and Investor Relations
+44 (0) 207 495 2379
e-mail: [email protected]
Investors:
Irina Koffler
LifeSci Advisors, LLC
+1 646 970 4681
[email protected]
1 https://www.pnas.org/doi/10.1073/pnas.2220272120
2 https://www.pnas.org/doi/10.1073/pnas.2309221120
A photograph accompanying this announcement is available at



